 The stability of the surrounding crystal structure guarantees that the neighboring atoms will not simply collapse around the vacancy. If a neighboring atom moves to occupy the vacant site, the vacancy moves in the opposite direction to the site which used to be occupied by the moving atom. Vacancy defects are lattice sites which would be occupied in a perfect crystal, but are vacant.These are frequently specified using Kröger–Vink notation. These dislocations permit ionic transport through crystals leading to electrochemical reactions. For historical reasons, many point defects, especially in ionic crystals, are called centers: for example a vacancy in many ionic solids is called a luminescence center, a color center, or F-center. Larger defects in an ordered structure are usually considered dislocation loops. However, these defects typically involve at most a few extra or missing atoms. Strict limits for how small a point defect is are generally not defined explicitly. They are not extended in space in any dimension. Point defects are defects that occur only at or around a single lattice point. Topological homotopy establishes a mathematical method of characterization. Several types of defects are often characterized: point defects, line defects, planar defects, bulk defects. The positions and orientations of particles, which are repeating at fixed distances determined by the unit cell parameters in crystals, exhibit a periodic crystal structure, but this is usually imperfect. Ī crystallographic defect is an interruption of the regular patterns of arrangement of atoms or molecules in crystalline solids. Made up by the presence of the required number of Fe 3+ ions.Disruption of the periodicity of a crystal lattice Electron microscopy of antisites (a, Mo substitutes for S) and vacancies (b, missing S atoms) in a monolayer of molybdenum disulfide. In crystals ofįeO some Fe 2+ cations are missing and the loss of positive charge is It mayĪctually range from Fe 0.93O to Fe 0.96O. Is FeO which is mostly found with a composition of Fe 0.95O. Many solids which are difficult to prepare in the stoichiometric compositionĪnd contain less amount of the metal as compared to the stoichiometricĭefect is mainly shown by transition elements. The electrical neutrality is maintained by acquiring a higher positive charge by the adjacent ion. The non-stoichiometric compounds may have metal deficiency due to the absence of the metal ion from its lattice site. 
Metal Deficiency Defect Due to Cationic Vacancies: Of an electron in interstitial space, the electrical conductivity of ZnO Sites and the electrons to neighbouring interstitial sites. The excess Zn 2+ ions move to interstitial Yellow. Now there is an excess of zinc in the crystal and its formulaīecomes Zn 1+xO. Stoichiometric Defects in Crystal Structure: Point or an atom in a crystalline substance. Even these crystals are not free of defects.ĭefects are the irregularities or deviations from ideal arrangement around a This happens when the crystallization process occurs at the fast or moderate rate. Single crystals are formed when the process of crystallization occurs at an extremely slow rate. These small crystals have defects in them. Usually a solid consists of an aggregate of a large number of small crystals. These imperfections in crystal lead to modification of some properties of the solid or may give rise to new properties. If the defects extend along a plane they are called plane defects. The line defects are the irregularities or deviations from an ideal arrangement in entire rows of lattice points. If lattice imperfections extend along the line they are called line defects. If the deviation from periodicity extends over microscopic regions of crystal then they are called lattice imperfections. Irregularities or deviations from ideal arrangement around a point or an atom Such defects are referred as point defects. Point defects are the They are due to missing or misplaced ions. 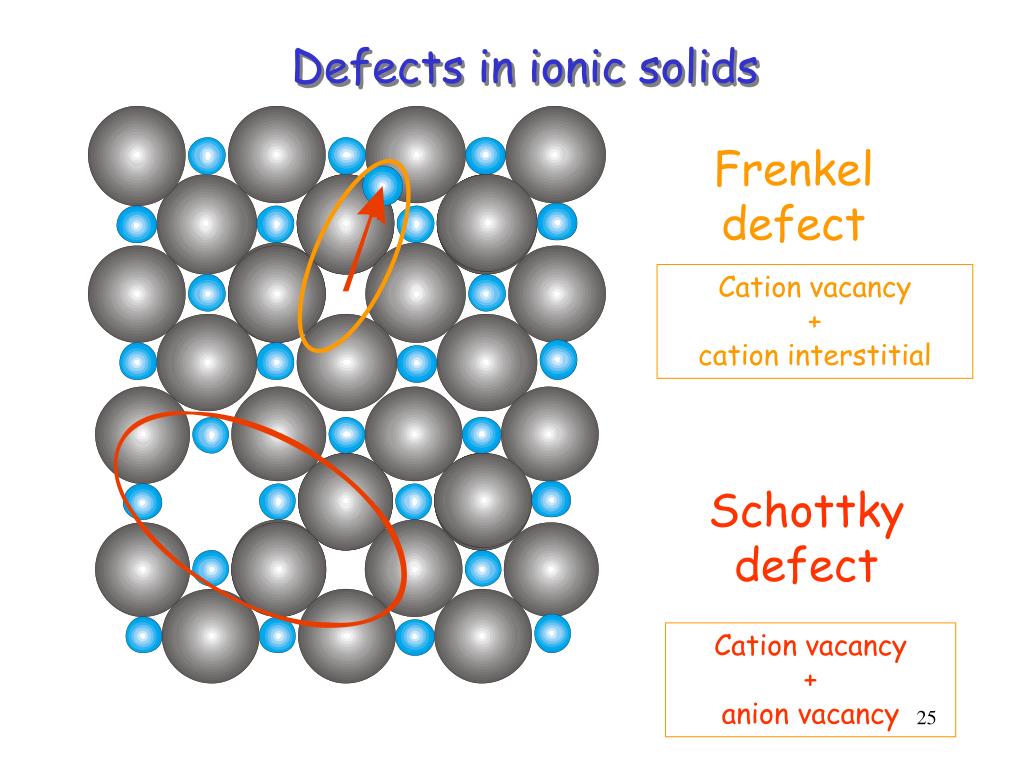
In the crystal due to the irregularities in the arrangement of atoms or ionsĪre called atomic imperfections. Such defects which are due to temperature change are referred as thermodynamic defects. The imperfection also may be due to impurities present in solid. 
Any deviation from the perfectly ordered arrangement constitutes a defect or imperfection. As the temperature increases, there is a change in the orderly arrangement of constituents in the crystal. The defects in crystal structure are basically irregularities in the arrangement of constituent particles. At absolute zero, crystals tend to have a tendency to have a perfectly ordered arrangement. This arrangement at absolute zero represents the lowest energy state of the crystal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
